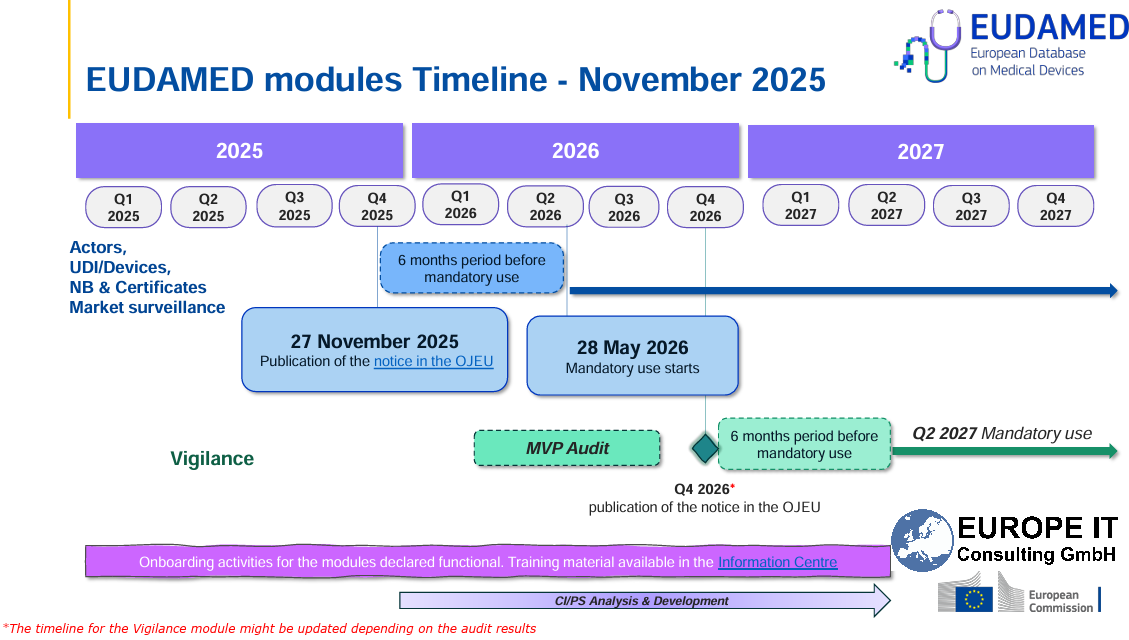
Current compliance deadlines
Stay compliant: The mandatory use of EUDAMED (EU) for registering medical devices is getting closer. Please note the current transition deadlines:
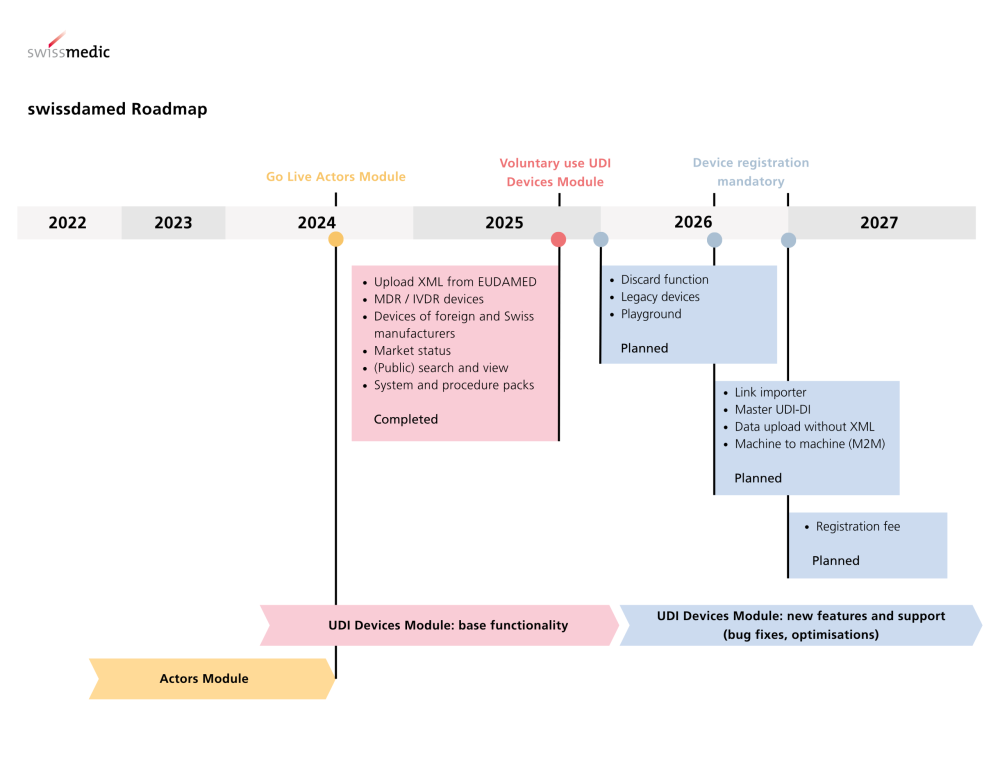
In Switzerland, too, many stakeholders are already subject to mandatory registration via swissdamed (CH). The parallel requirements call for an integrated strategy:
GUDI helps you reliably meet both deadlines through automated processes directly in your SAP system.